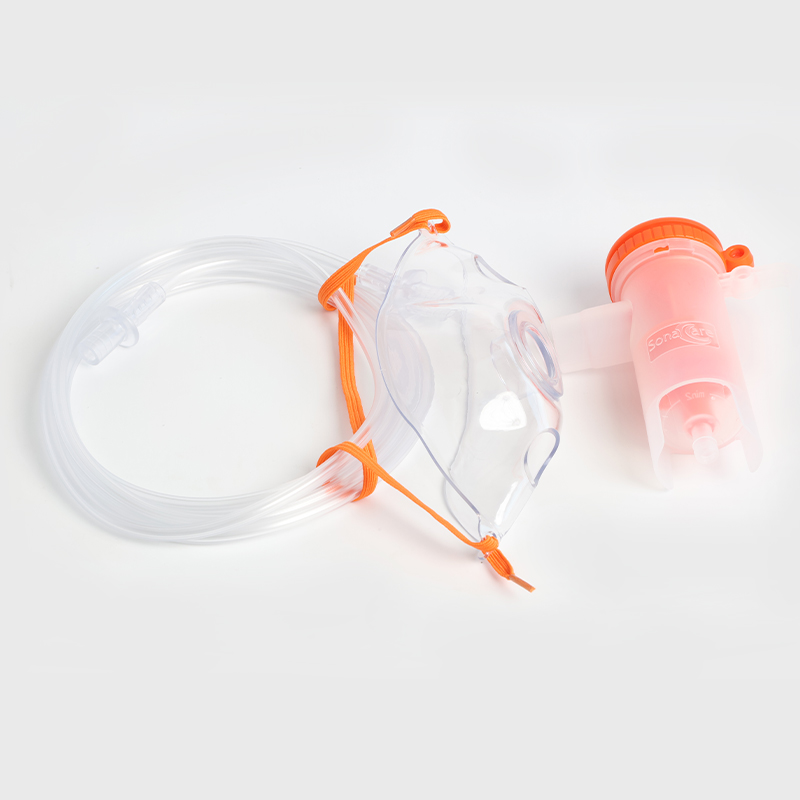
Culture interpretation time:
30 minutes: Hydrogen peroxide indicator type yields results in 30 minutes.
1 hour: Steam indicator type yields results in 60 minutes.
3 hours: Steam indicator type yields results in 180 minutes.
Data processing and storage:
After the culture is completed, the reader automatically saves the culture results, which can be easily
Viewed on the device. The data can also be exported through the USB interface or Ethernet interface on the device, allowing users to query, edit, and print historical records on other devices.
Culture Information Traceability System:
During the culture process, relevant information such as the indicator and operator can be inputted. This information can be included in the saved final culture results and can be queried, edited, and printed
through the "Data Processing and Storage" feature mentioned earlier.
Note: Culture traceability information can only be entered after the culture with the indicator has started.
Visualization:
The reader adopts a touchscreen for human-machine interaction, supporting a visual graphical user interface.
Multi-directional Alarm Prompts:
The Automatic Biological Reader provides audio and visual alarm prompts, allowing operators to have comprehensive and real-time information about the culture process.



 English
English Español
Español Français
Français



























 CONTACT US
CONTACT US