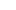How can optical trocars improve the accuracy of abdominal wall puncture during surgery?
Content
- 1 Optical Trocars Reduce Blind Entry Injuries by Providing Real-Time Visualization of Each Tissue Layer During Puncture
- 2 How Optical Trocars Work: The Mechanism Behind Visualized Entry
- 3 Clinical Evidence: What the Data Shows About Optical Trocar Safety
- 4 Patient Populations Where Optical Trocars Provide the Greatest Accuracy Benefit
- 5 Optical Trocar vs. Alternative Entry Techniques: A Direct Comparison
- 6 Surgical Technique: Step-by-Step Optical Trocar Insertion for Accurate Abdominal Entry
- 7 Optical Trocar Selection: Key Specifications That Affect Puncture Accuracy
- 8 Port Site Hernia Risk: Why Dilating Optical Trocars Offer a Structural Advantage
- 9 Frequently Asked Questions About Optical Trocars in Laparoscopic Surgery
- 9.1 Q1: Does using an optical trocar require pre-established pneumoperitoneum?
- 9.2 Q2: How does the learning curve for optical trocar use compare to conventional entry techniques?
- 9.3 Q3: Are optical trocars suitable for all laparoscopic procedures, or are there situations where they should not be used?
- 9.4 Q4: What should the surgeon do if tissue planes are not clearly identifiable during optical trocar insertion?
- 9.5 Q5: How do optical trocars perform specifically in patients with morbid obesity (BMI ≥ 40 kg/m²)?
- 9.6 Q6: What is the cost difference between optical and conventional trocars, and is the additional cost justified?
Optical Trocars Reduce Blind Entry Injuries by Providing Real-Time Visualization of Each Tissue Layer During Puncture
Optical trocars improve abdominal wall puncture accuracy by enabling the surgeon to visualize each tissue layer—skin, subcutaneous fat, anterior fascia, muscle, posterior fascia, and peritoneum—in real time as the trocar advances, eliminating the blind force-based entry that causes the majority of laparoscopic access injuries. Clinical data published in surgical journals consistently shows that optical entry techniques reduce major vascular injury rates to below 0.02% and bowel injury rates to below 0.04%, compared to 0.05–0.3% for conventional blind Veress needle or non-optical trocar techniques in high-risk patient populations.
Access-related complications—including vascular, bowel, and solid organ injuries during trocar insertion—account for approximately 50% of all major laparoscopic complications and up to 40% of laparoscopy-related deaths. The optical trocar addresses the fundamental cause of these injuries: the inability to see what lies immediately ahead of the tip during blind mechanical entry. This article examines the mechanism, clinical evidence, technique, and practical selection criteria that allow surgeons to use optical trocars effectively.
How Optical Trocars Work: The Mechanism Behind Visualized Entry
An optical trocar integrates a transparent, conical or pyramidal obturator tip with a central optical channel that accepts a 0° or 30° laparoscope. As the surgeon applies rotational or axial force to advance the trocar, the laparoscope simultaneously transmits a live magnified image of the tissue immediately in front of the tip. The surgeon observes each fascial and muscular layer being progressively separated—rather than cut—as the trocar advances under direct vision.
Key Structural Components of an Optical Trocar
- Transparent dilating obturator: Made from optical-grade polycarbonate or PETG, the obturator tip allows the laparoscope lens to remain in contact with the tissue face throughout insertion. The clarity of this component directly determines image quality—medical-grade optical trocars maintain transparency to within 3–5 mm of the tip edge.
- Trocar cannula with gas-tight seal: The working channel that maintains pneumoperitoneum after entry. Available in 5 mm, 10 mm, 12 mm, and 15 mm diameters to accommodate different laparoscopic instrument sizes.
- Retractable blade safety mechanism (in some designs): Certain optical trocars combine the visual channel with a spring-loaded blade that retracts immediately after the peritoneum is penetrated, providing a secondary mechanical safety layer against plunge injury.
- Threaded or smooth cannula body: Threaded cannulas provide greater resistance to accidental dislodgement during the procedure—particularly important in obese patients where the abdominal wall thickness reduces the mechanical grip of smooth-bodied cannulas.
The Tissue Layer Progression Visible During Optical Entry
During optical trocar insertion, a trained surgeon identifies the following sequential landmarks on the laparoscopic image, each confirming correct progression through the abdominal wall:
- Subcutaneous adipose tissue: Yellow, lobular fat tissue with visible fibrous septa. Rotational insertion allows progressive dilation without cutting.
- Anterior rectus sheath / external oblique fascia: White, dense fibrous tissue with a crosshatch fiber pattern. Increased resistance is palpated and confirmed visually before penetration.
- Rectus abdominis or oblique muscle: Red-pink striated tissue with visible muscle fiber bundles. Blood vessels within the muscle belly are visible and avoidable.
- Posterior rectus sheath / transversalis fascia: Another fibrous white layer—the last anatomical barrier before the peritoneal cavity.
- Preperitoneal fat (if present): Yellow loose areolar tissue confirming proximity to peritoneum—visible in most patients above the arcuate line.
- Peritoneum: Glistening, semi-transparent membrane—its visual appearance confirms imminent entry into the peritoneal cavity. The surgeon can pause here to confirm no underlying adhesions or viscera are visible before completing entry.
Clinical Evidence: What the Data Shows About Optical Trocar Safety
The safety advantage of optical trocar entry is supported by multiple published clinical series and meta-analyses. The following data points represent peer-reviewed findings:
| Entry Technique | Major Vascular Injury Rate | Bowel Injury Rate | Failed Entry Rate | Conversion to Open Rate |
|---|---|---|---|---|
| Veress needle (standard) | 0.05–0.10% | 0.08–0.15% | 1.0–3.5% | 0.3–0.8% |
| Non-optical bladed trocar | 0.04–0.08% | 0.05–0.10% | 0.8–2.0% | 0.2–0.5% |
| Optical trocar (dilating) | 0.01–0.02% | 0.02–0.04% | 0.3–0.8% | 0.05–0.15% |
| Hasson open technique | 0.02–0.05% | 0.03–0.07% | 0.1–0.5% | 0.2–0.6% |
A 2019 systematic review in Surgical Endoscopy analyzing over 350,000 laparoscopic entries found that optical trocar entry reduced major access complications by approximately 60% compared to standard Veress needle technique in patients with prior abdominal surgery—the highest-risk population for access injuries due to intra-abdominal adhesions. In standard-risk patients, the safety difference between entry methods narrows, but the optical trocar consistently demonstrates the lowest major injury rates across all published series.
Figure 1: Comparison of vascular injury, bowel injury, and failed entry rates across laparoscopic access techniques
Patient Populations Where Optical Trocars Provide the Greatest Accuracy Benefit
While optical trocars improve puncture accuracy in all laparoscopic cases, the benefit is most clinically significant in patients where anatomical variability or prior surgical history increases the complexity of blind entry:
Obese Patients (BMI ≥ 30 kg/m²)
In obese patients, the abdominal wall thickness may exceed 8–15 cm, and the anatomical landmarks used during blind Veress needle insertion are obscured or displaced. The umbilicus—the standard Veress insertion point—may be located significantly inferior to the aortic bifurcation in obese patients, increasing major vessel risk during straight-axis insertion. Optical trocar entry allows the surgeon to navigate the thick adipose layer under direct vision, identify the fascial layers despite poor landmark definition, and confirm peritoneal entry at the correct depth—independent of surface anatomical landmarks.
Patients with Prior Abdominal Surgery
Previous laparotomy, Pfannenstiel incisions, or prior laparoscopy creates peritoneal adhesions in an estimated 50–90% of patients, depending on the extent of prior surgery. Blind entry in the presence of adhesions risks piercing bowel or omentum adherent to the anterior abdominal wall at the insertion site. Optical entry allows the surgeon to identify and avoid adhesions at the peritoneal surface before completing penetration—pausing at the preperitoneal layer to assess the peritoneal reflection for underlying structures before final entry.
Pediatric Patients
In pediatric laparoscopy, the thin abdominal wall and reduced peritoneal cavity depth significantly reduce the safety margin during blind entry. The distance from umbilical skin to the aortic bifurcation in a child weighing 10 kg is approximately 2.5–4.0 cm—compared to 7–12 cm in an average adult. Optical trocars provide real-time depth confirmation in this compressed anatomical space, allowing controlled millimeter-by-millimeter entry that blind techniques cannot achieve.
Patients with Abdominal Wall Abnormalities
Patients with umbilical hernia repair mesh, diastasis recti, or prior abdominal wall reconstruction present atypical tissue planes that can mislead both tactile sensation and CO₂ insufflation pressure readings during blind Veress entry. Optical visualization of actual tissue layers—regardless of their abnormal appearance—allows accurate identification of the peritoneum even when standard anatomical landmarks are absent or distorted.
Optical Trocar vs. Alternative Entry Techniques: A Direct Comparison
| Criterion | Optical Trocar | Veress Needle + Blind Trocar | Hasson Open Entry |
|---|---|---|---|
| Real-time visualization | Yes — continuous during insertion | No — blind insertion | Partial — direct open view only |
| Incision size required | 5–12 mm (trocar-matched) | 5–12 mm | 20–30 mm (open cutdown) |
| Entry time | 2–5 minutes | 3–8 minutes (including insufflation) | 5–15 minutes |
| Performance in obese patients | Reliable; visual navigation through fat | Difficult; high failed entry rate | Technically challenging; long incision |
| Performance in prior surgery | Good; adhesions visible before entry | High risk; adhesions undetectable | Preferred; direct adhesiolysis possible |
| Pneumoperitoneum required before entry | No; can enter without pre-insufflation | Yes; requires Veress insufflation first | No; open technique |
| Tissue trauma | Minimal; dilating non-cutting tip | Moderate; sharp cutting tip | Greater; full thickness incision |
| Port site hernia risk | Lower; dilated fascia (no cutting) | Moderate; cut fascial defect | Higher; larger fascial defect |
Surgical Technique: Step-by-Step Optical Trocar Insertion for Accurate Abdominal Entry
The accuracy benefit of an optical trocar is fully realized only when the insertion technique is executed correctly. The following sequence reflects established laparoscopic surgical practice:
- Patient positioning and abdominal wall elevation: Position the patient supine with a slight Trendelenburg tilt if required. Have the assistant elevate the abdominal wall manually or with towel clips to increase the distance between the anterior abdominal wall and underlying viscera—reducing organ proximity during entry.
- Skin incision: Make a skin incision precisely matching the outer diameter of the trocar cannula (typically 10–12 mm for a standard optical trocar). An incision that is too small creates excessive resistance that obscures the tissue plane image by pressing the obturator face against compressed tissue.
- Laparoscope insertion and white balance: Insert the laparoscope into the optical channel of the obturator and perform white balance against a white gauze. Confirm that the image is clear and focused before beginning insertion—a fogged or out-of-focus image from the start negates the visualization advantage.
- Insertion with rotational advance: Apply gentle rotational pressure (clockwise-counterclockwise oscillation) rather than direct axial thrust. Rotational insertion dilates tissue fibers rather than cutting them, and requires 20–40% less force than direct push techniques—reducing the risk of uncontrolled plunge after fascial breakthrough.
- Layer-by-layer identification: Advance the trocar only as fast as the image updates can be processed—typically 2–5 mm per second. Identify each tissue layer (subcutaneous fat, anterior fascia, muscle, posterior fascia, preperitoneal fat, peritoneum) before advancing through it.
- Peritoneal entry confirmation and pause: When the glistening peritoneum becomes visible, pause insertion. Inspect the peritoneal surface for adhesions, bowel, or vessels before completing entry. This 5–10 second pause is the most important safety step in the optical entry sequence.
- Pneumoperitoneum establishment: After confirmed peritoneal entry, connect the CO₂ insufflation line and establish pneumoperitoneum to 12–15 mmHg before removing the obturator and proceeding with laparoscopy.
Optical Trocar Selection: Key Specifications That Affect Puncture Accuracy
Not all optical trocars perform equally. The following specifications determine whether a given device delivers the visualization quality needed for accurate abdominal wall puncture:
- Obturator tip transparency grade: Optical-grade polycarbonate provides the clearest image transmission. Lower-grade plastics develop micro-scratches within a single procedure that degrade image clarity. For single-use optical trocars, verify that the transparency grade meets ISO 10993 biocompatibility standards and that the optical channel maintains ≥85% light transmission from a standard 10mm laparoscope.
- Obturator tip geometry (dilating vs. bladed): Purely dilating conical tips provide better visualization (the tissue face remains flat against the lens) and cause less tissue trauma than hybrid dilating-bladed designs. Bladed tips cut rather than spread, which can make tissue plane identification less distinct on the image—though they require less insertion force in dense fibrous fascia.
- Laparoscope compatibility: Confirm that the optical channel inner diameter matches the laparoscope being used (10mm optical channel for standard 10mm laparoscope; 5mm channel for 5mm laparoscope). A loose fit allows the laparoscope to shift off-axis during insertion, degrading image centering and potentially scratching the obturator interior.
- Cannula thread design: For procedures in obese patients or where the trocar will remain in place throughout a long procedure, threaded cannulas reduce the frequency of accidental trocar dislodgement. Studies show threaded cannulas have a 3–5 times lower dislodgement rate in patients with abdominal wall thickness above 5 cm compared to smooth-bodied cannulas.
- Single-use vs. reusable: Single-use optical trocars guarantee consistent obturator tip sharpness and optical clarity at every procedure. Reusable devices can be more economical but require strict reprocessing protocols—optical-grade obturators that are steam-autoclaved more than 20–30 times show measurable degradation in tip geometry and optical transmission that can compromise visualization quality.
Figure 2: Estimated distribution of optical trocar tip designs used in laparoscopic surgery
Port Site Hernia Risk: Why Dilating Optical Trocars Offer a Structural Advantage
Port site hernias (PSH) are a recognized complication of laparoscopic surgery, occurring at fascial defects created by trocar insertion. The incidence of port site hernia at 10–12 mm trocar sites is reported at 0.65–2.8% in published series—with the highest rates at umbilical ports and in obese patients. The dilating mechanism of optical trocars provides a structural advantage that reduces this complication rate:
A dilating optical trocar separates fascial fibers radially rather than cutting them. When the trocar is removed at procedure completion, the dilated fascial fibers partially reapproximate under their natural elastic tension—leaving a smaller effective defect than a cut fascia of the same diameter. Studies comparing dilating versus bladed 12 mm ports show a port site hernia rate of 0.3% for dilating trocars versus 1.2–1.8% for cutting trocars at the same port size—a 4 to 6-fold difference that represents a meaningful reduction in long-term reoperation risk, particularly for commonly used 12 mm trocar sites.
Frequently Asked Questions About Optical Trocars in Laparoscopic Surgery
Q1: Does using an optical trocar require pre-established pneumoperitoneum?
No—this is one of the practical advantages of optical trocars over conventional Veress needle technique. Optical trocars can be inserted directly through the abdominal wall without prior pneumoperitoneum. The surgeon visualizes tissue layers under direct pressure from the obturator tip, without relying on intraperitoneal gas distension to create a safety buffer. However, some surgeons prefer to establish a small initial pneumoperitoneum (typically 8–10 mmHg) using a Veress needle at an alternative site before optical trocar insertion, particularly in patients with suspected dense adhesions—the partial distension helps separate the bowel from the anterior wall at the insertion point. Either approach is valid; the published clinical data shows no significant safety difference between direct optical entry and optical entry after pre-insufflation in standard-risk patients.
Q2: How does the learning curve for optical trocar use compare to conventional entry techniques?
The learning curve for optical trocar insertion is generally considered shorter than for Veress needle technique, primarily because visual feedback accelerates skill acquisition. Published data from surgical training programs suggests that residents reach consistent tissue layer identification accuracy after 15–25 supervised optical trocar insertions, compared to 30–50 procedures for reliable Veress needle technique based on tactile and pressure feedback alone. The key learning elements for optical entry are: recognizing the visual appearance of each tissue layer, maintaining a steady insertion speed that allows image processing, and developing the judgment to pause appropriately at the peritoneum before completing entry. Structured simulation training on abdominal wall models significantly accelerates tissue layer recognition before clinical application.
Q3: Are optical trocars suitable for all laparoscopic procedures, or are there situations where they should not be used?
Optical trocars are appropriate for the majority of elective laparoscopic procedures. There are a small number of situations where alternative entry strategies should be considered: (1) Emergency laparoscopy for trauma where speed is paramount and the surgical team is not routinely trained in optical entry—in these circumstances, Hasson open entry by an experienced surgeon may be faster and safer. (2) Patients with giant umbilical hernia or complete loss of abdominal wall anatomy at the intended entry site—where the normal tissue plane sequence may not be identifiable even optically. (3) Extremely thin patients (BMI below 18 kg/m²) where the minimal subcutaneous tissue makes the insertion distance to the peritoneum very short—increasing the risk of bowel contact even with the controlled insertion speed required for optical entry. In none of these situations is the optical trocar contraindicated by device design; the decision is based on individual surgical judgment and team training status.
Q4: What should the surgeon do if tissue planes are not clearly identifiable during optical trocar insertion?
If tissue layer visualization becomes unclear during insertion—due to fogging, blood on the lens surface, or atypical tissue anatomy—the correct response is to stop insertion immediately, withdraw the trocar 5–10 mm, and reassess before continuing. Causes of poor visualization include: condensation on the laparoscope tip (correctable by warming the scope tip in warm saline before insertion), blood obscuring the lens (requires trocar withdrawal and scope cleaning), or tissue architecture that does not match the expected pattern (should prompt reassessment of the insertion site or conversion to Hasson technique if anatomy is uncertain). Never advance the trocar while the image is unclear—the primary benefit of optical entry is eliminated if insertion continues without visualization. If visualization cannot be reliably maintained, conversion to Hasson open entry at the same or an alternative site is appropriate and should not be considered a failure of the technique.
Q5: How do optical trocars perform specifically in patients with morbid obesity (BMI ≥ 40 kg/m²)?
In morbid obesity, optical trocars perform well but require specific technique modifications. The major challenge is that the extended insertion distance through a thick adipose layer means the laparoscope's working distance is stretched, reducing image magnification and clarity at the deep tissue layers. Practical adaptations include: using a 30° angled laparoscope rather than 0° to improve viewing angle in the compressed tissue face; applying additional abdominal wall elevation (lifting the pannus manually or using a lift device) to decompress the adipose layer before insertion; and selecting a trocar with a longer shaft (available in 100–150 mm lengths for obese patients versus the standard 70–80 mm) to ensure adequate cannula reach into the peritoneal cavity after entry. With these adaptations, published bariatric surgery series report optical entry success rates of 96–99% in patients with BMI 40–60 kg/m², with major access complication rates below 0.05%.
Q6: What is the cost difference between optical and conventional trocars, and is the additional cost justified?
Single-use optical trocars cost approximately $45–$120 per unit depending on diameter and manufacturer, compared to $8–$25 for a standard single-use bladed trocar—a cost premium of $30–$100 per port site. For a typical 4-port laparoscopic procedure using one optical primary port, the additional cost is $30–$100 per procedure. This cost is placed in a different perspective when considered against the consequences of a major access injury: management of a major vascular injury from trocar insertion requires emergency conversion, ICU admission, and vascular repair—average costs of $25,000–$80,000 per event in published healthcare economics analyses, excluding medicolegal costs. Even at a conservatively estimated 60% reduction in major access injury rate, the break-even calculation at a facility performing 500 laparoscopic procedures annually is reached at an injury rate of approximately 0.02%—consistent with published Veress needle complication rates in high-risk populations. For high-risk patient categories (obese, prior abdominal surgery), the economic case for routine optical trocar use as the primary port is generally considered favorable by published health technology assessment analyses.



 English
English Español
Español Français
Français










 CONTACT US
CONTACT US